Bird saliva is collected using sterile swabs (usually synthetic fiber with a plastic shaft) wiped along the inside of the bird's mouth and throat, then placed into a small tube of transport media like BHI broth or viral transport medium. That's the core method used by vets, wildlife biologists, and diagnostic labs worldwide. Everything else in this guide is about doing it right, staying safe, and knowing when to hand the job off to someone with the proper tools.
How Is Bird Saliva Collected: Safe Methods and Next Steps
Why anyone would collect bird saliva in the first place
Most people land on this topic from one of three directions: they had a close encounter with a bird (a bite, a peck, or a face full of bird sneeze) and want to know if they were exposed to something dangerous; they keep pet birds and a vet has asked for a sample; or they're involved in backyard flock management or wildlife work and need to test for avian influenza or Newcastle disease. Less commonly, researchers collect oral secretions for studies on bird digestive enzymes, saliva composition, or disease surveillance in wild populations.
The health concern angle is real. Bird oral secretions can carry pathogens including avian influenza A viruses, Chlamydia psittaci (which causes psittacosis in humans), and Salmonella, among others. If you are looking for what bird dust is, it helps to understand that dried or aerosolized particles from birds can still contain infectious material Bird oral secretions can carry pathogens. If you've been bitten by a bird or had contact with its secretions and you're worried about exposure, that context shapes what kind of sample matters and who needs to collect it. For most people reading this after a bird bite or a cleanup incident, the question isn't really about collecting saliva from the bird but rather about whether they were exposed to something. Those are related but different problems.
What you can do at home versus what needs a lab
Here's the honest breakdown: at home, without proper materials, you can collect a rough oral swab from a pet bird or capture visible secretions on a clean surface for basic observation. What you cannot reliably do at home is collect a sample in a way that will hold up diagnostically. The swab material matters, the transport medium matters, the temperature during storage matters, and the way you package it for shipping matters. Miss any of those and you may end up with a result that says nothing useful, or worse, a false negative on something you really needed to detect.
That said, knowing the difference between a rough at-home collection and a proper lab submission helps you decide which path you're on. If you're just trying to capture some visible drool or secretion from a bird's beak for general observation or a basic vet exam, a clean cotton swab and a zip-lock bag gets you there. If you need PCR-quality results for pathogen detection, you need the right synthetic swab, the right media, cold storage, and a submission process through a veterinary diagnostic lab.
The main collection methods, from field work to the clinic
Oropharyngeal (oral-pharyngeal) swabbing

This is the gold standard for collecting bird oral secretions for diagnostic testing. A sterile synthetic swab (Dacron or flocked fiber, plastic shaft) is inserted into the bird's open mouth and rotated against the back of the throat and oral cavity to absorb secretions. USGS field teams use this method for avian influenza surveillance, and both USDA APHIS and multiple state veterinary diagnostic labs (Iowa State, Texas A&M TVMDL, NDSU, and others) specify oropharyngeal swabs as the preferred collection route for gallinaceous poultry like chickens and turkeys. For domestic waterfowl, cloacal swabs are often preferred instead, since waterfowl shed influenza virus more heavily from the gastrointestinal tract.
Tracheal swabbing
In clinical veterinary settings, a swab is sometimes passed deeper into the trachea to collect lower airway secretions. This is more invasive, requires proper restraint and ideally sedation for smaller birds, and is typically done by a vet or trained technician. It's not something to attempt at home.
Secretion capture from surfaces or ropes
For situations where directly swabbing a bird isn't feasible (for example, collecting from a flock rather than individual birds), labs have documented a clever workaround: a cotton rope or sock is hung in the pen, birds mouth and peck at it naturally, and the rope is then collected and submitted as a pooled oral fluid sample. University of Minnesota's VDL specifically notes cotton as a material that holds the most oral secretions in this context. This works at scale for flock surveillance but requires the same careful handling and transport as direct swab collection.
Drool and visible secretion capture

Some birds produce visible oral secretions, particularly when ill, stressed, or regurgitating. These can be captured with a swab or absorbent material directly from the beak or nearby surfaces. The same material rules apply: synthetic swabs hold up better than cotton-tipped wooden sticks for molecular diagnostics, and anything you collect needs to go into appropriate transport media quickly to preserve the sample.
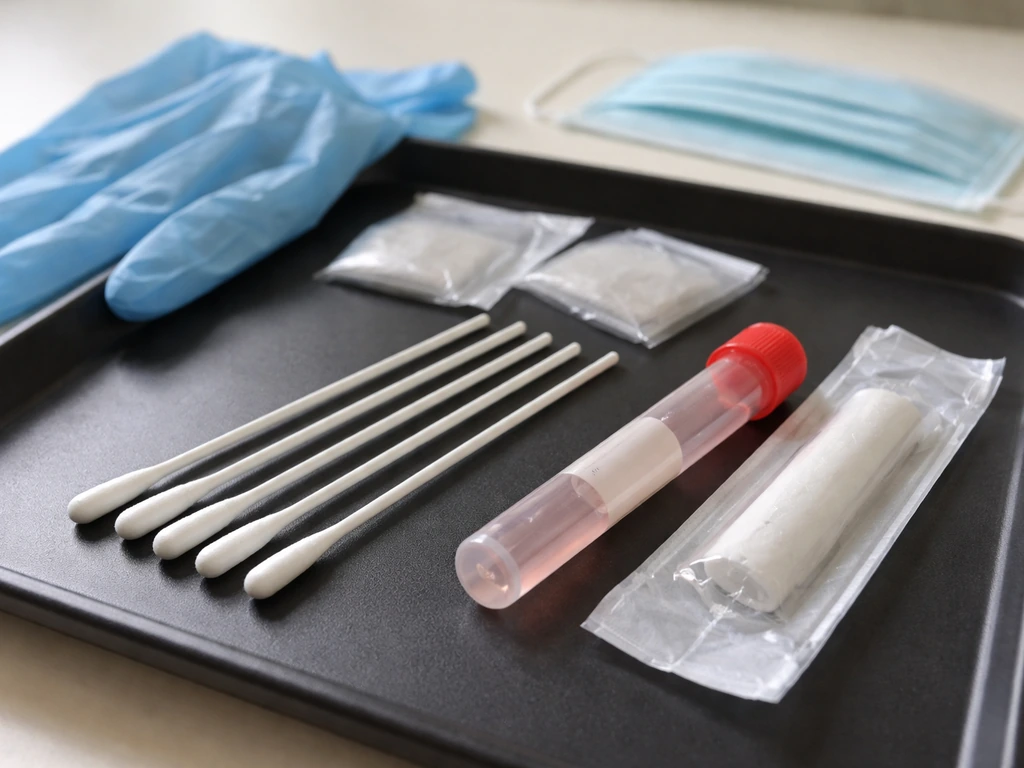
The right materials matter more than you'd think
This is where a lot of home collection attempts fall apart. Different labs have clear lists of what they won't accept. Calcium alginate swabs are explicitly listed as unacceptable by the UK VDL and Texas DSHS, among others. Cotton-tipped swabs with wooden shafts are also flagged as not recommended for molecular work (PCR testing) because wooden shafts and cotton fibers can inhibit the assay. The go-to choice is a synthetic fiber swab (Dacron or flocked nylon) on a plastic or wire shaft.
| Swab Type | Acceptable for PCR/Molecular? | Notes |
|---|---|---|
| Synthetic (Dacron/flocked), plastic shaft | Yes | Preferred by most VDLs including NDSU, TVMDL, and UK VDL |
| Cotton tip, plastic shaft | Sometimes acceptable | Holds more fluid but may interfere with some molecular assays; UMN notes cotton holds oral fluid well for oral fluid submissions |
| Cotton tip, wooden shaft | Not recommended | Wood and cotton can inhibit PCR; avoid for diagnostic submissions |
| Calcium alginate swab | No | Explicitly unacceptable; listed on UK VDL, Texas DSHS, and CDC guidance |
Handling, storage, and transport: this is where most samples fail
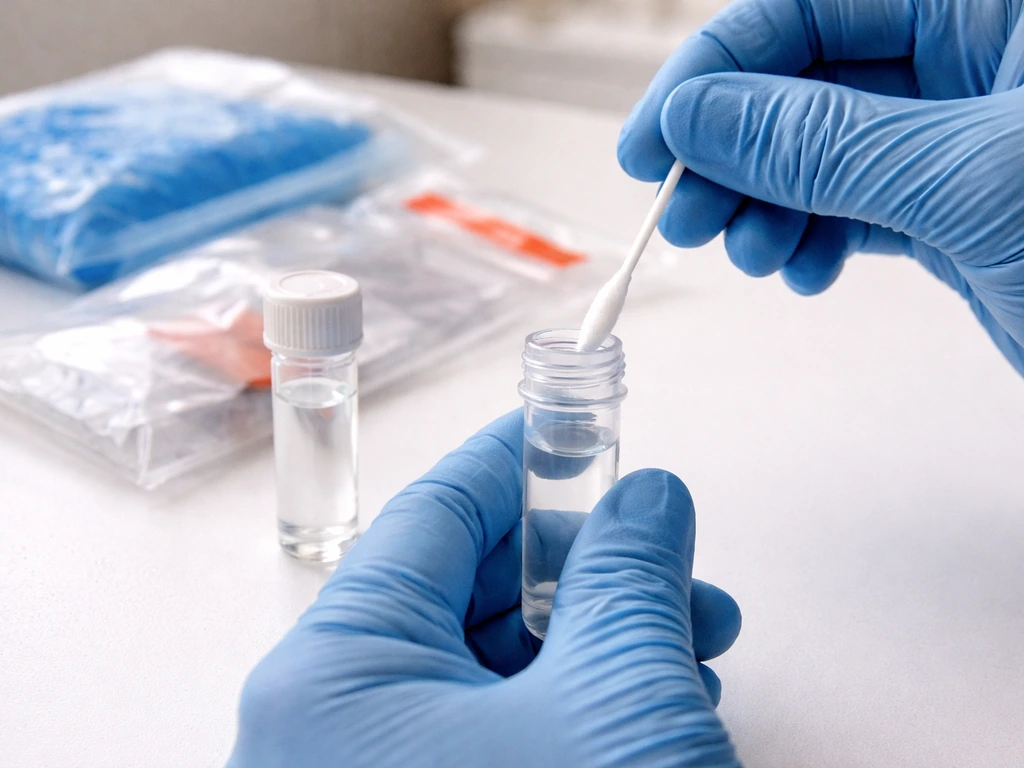
Once you've collected the swab, it needs to go into transport media immediately. The two most commonly specified options are BHI broth (Brain Heart Infusion) and viral transport medium (VTM) or universal transport medium (UTM). Iowa State VDL specifies a minimum of 3 mL of BHI broth for avian influenza swab pools. CDC protocols specify 1 to 3 mL of VTM per swab. The media isn't optional: it keeps the virus or pathogen viable during transport. A dry swab sitting in an empty red-top tube, as NDSU notes, is acceptable at best and not ideal, meaning you may get a degraded or inconclusive result.
Pooling has rules too. Iowa State's submission guidance specifies a maximum of five swabs pooled into a single tube. Going beyond that dilutes the sample enough to cause false negatives, especially for PCR-based tests.
Temperature is critical. Samples stored and shipped at 2 to 8 degrees Celsius maintain diagnostic quality for short transport windows. If there's any chance of a delay beyond a few days, samples should be frozen at minus 70 degrees Celsius or below (not a standard home freezer, which only reaches about minus 18 to minus 20 degrees). Research comparing transport conditions confirmed that breaking the cold chain directly degraded PCR detection performance for avian influenza and Newcastle disease virus. If you don't have a cold chain you trust, your sample is already compromised.
Labeling and packaging are the final piece. Each tube should be labeled with the bird species, collection date, collection site (beak/oropharyngeal vs. cloacal), and your contact information. Tubes go into a secondary leak-proof container with absorbent material around them, then into a rigid outer container, following the triple-packaging standard used for potentially infectious biological material. USDA APHIS and Oregon State's OVDL both specify this triple-layer approach for shipping. This packaging is also a legal requirement for shipping regulated biological material, not just a best practice.
Stay safe: PPE and contamination control
Bird oral secretions can carry the same pathogens as bird droppings, and in some cases higher concentrations. Avian influenza virus, Chlamydia psittaci, Salmonella, and other organisms can all be present. Treat every collection as if the bird is shedding something. That means gloves (nitrile, not latex if you can help it), eye protection or a face shield if there's any chance of the bird flapping or spraying, and a mask. The N95 level is appropriate if you're working with birds that may have respiratory illness or are part of a suspected HPAI outbreak.
After collection, wash hands thoroughly with soap and water for at least 20 seconds. Any surfaces the bird or the sample came into contact with should be disinfected with a bleach-based solution (a 1:10 dilution of household bleach in water works for most surfaces). The outside of the sample tube should be treated as contaminated and wiped down before handling further. CDC and BMBL biosafety guidance both stress that specimen containers should be handled as potentially infectious on their exterior surfaces.
Avoid generating aerosols. Opening tubes, sneezing near samples, or agitating liquid samples without a lid can send infectious particles airborne. In a diagnostic lab this is handled with biosafety cabinets. At home, it means working carefully, outdoors or in good ventilation, and not leaning over open tubes.
- Wear nitrile gloves, eye protection, and a mask during collection and handling
- Avoid touching your face until gloves are off and hands are washed
- Disinfect any surface the bird or sample contacted using a bleach-based cleaner
- Wipe the outside of all sample tubes with disinfectant before storing or handing them off
- Don't open tubes unnecessarily or in enclosed spaces with poor ventilation
- Bag and seal used PPE before disposal
What can actually be tested from bird saliva
The main diagnostic targets for oropharyngeal swabs from birds are avian influenza A viruses (detected by rRT-PCR, specifically targeting the matrix gene), Newcastle disease virus, Chlamydia psittaci, and general bacterial cultures. PCR-based testing is far more sensitive than older methods and is the standard at state and federal veterinary diagnostic labs. If you are asking what bird spit tastes like, it is best to treat it as potentially infectious and not try to taste it. If your question is more about the human side, like why does your breath smell like bird seed after handling birds, the key is understanding exposure risk rather than collecting bird saliva for testing. For pet bird health assessments, a vet may use oral swabs to look for fungal infections like Candida or bacterial overgrowth.
The limits are worth understanding. Oral swabs are not a perfect substitute for tissue samples or full necropsy in a bird that has died. A live bird shedding at low levels may test negative even when infected, especially if the sample sat in a warm car for two hours before reaching the lab. Timing matters: swabs collected during active illness or early infection catch more virus than samples from birds that have partially recovered. And no matter how perfect your swab technique, the wrong transport media or a broken cold chain will undermine the result.
It's also worth noting that bird saliva composition, its enzymes, proteins, and microbial content, varies significantly by species. Bird saliva benefits are mostly tied to what labs can detect from oral secretions, so the sample quality and handling matter as much as the collection itself. What you can learn from a parrot's oral secretions is different from what's diagnostically useful from a wild mallard. If the context here is health risk after human contact with bird saliva or droppings, the answer is that most routine health screenings after bird exposure don't rely on collecting saliva from the bird at all; they focus on monitoring the exposed person.
When to stop DIY and call a vet or diagnostic lab
There are clear moments when home collection stops making sense and professional involvement becomes necessary. If you're dealing with a suspected avian influenza case (multiple birds dying quickly, especially if waterfowl or backyard poultry are involved), stop immediately and contact your state veterinarian or USDA APHIS. HPAI is a reportable disease and comes with specific official sampling and submission protocols that you are legally required to follow. Trying to self-collect and submit in that situation can delay official response and may not produce usable results anyway.
For pet birds showing signs of respiratory illness, lethargy, or unusual oral discharge, the right call is a vet visit. An avian vet can collect a proper oropharyngeal swab with appropriate restraint, submit it through an established lab relationship, and interpret results in clinical context. They'll also know which lab to use, what submission forms are required, and whether additional samples (blood, feces, tissue) are needed alongside the swab.
If you don't have access to viral transport medium or BHI broth, a properly validated synthetic swab, a reliable cold chain for shipping, or experience restraining birds safely, those are all reasons to stop and let a professional handle it. Using the wrong materials or breaking the cold chain doesn't just give you a bad result; it can give you a false reassurance or a false alarm, both of which have real consequences.
- Multiple birds dying or showing severe illness: contact your state vet or USDA APHIS immediately
- Suspected HPAI or Newcastle disease: do not self-submit; official sampling protocols apply
- Pet bird with respiratory symptoms or unusual oral discharge: see an avian vet
- You don't have synthetic swabs, proper transport media, or a cold chain: stop and get help
- You were bitten or had oral secretion exposure and are concerned about your own health: contact your doctor, not a bird lab
- Results will be used for any official or regulatory purpose: always go through a licensed vet or certified diagnostic lab
The practical upshot is this: for curiosity or a basic home health check on a pet bird, a simple swab is fine. People also use the phrase “<a data-article-id="225B9C73-319B-49B9-8634-B768F9D36E7D"><a data-article-id="225B9C73-319B-49B9-8634-B768F9D36E7D">dirty bird</a></a>” when discussing chickens and their oral secretions, which is why you may hear that nickname online. For anything that needs to count diagnostically, the collection method, materials, and chain of custody from swab to lab result have to meet professional standards. Cutting corners anywhere in that chain is the most common reason tests come back inconclusive or invalid, and that's a frustrating outcome when you needed a real answer.
FAQ
Can I collect bird saliva with a home cotton swab and still get reliable PCR results?
Not usually. For molecular diagnostics, cotton-tipped swabs and wooden shafts can inhibit PCR, and the sample often lacks the validated synthetic swab and transport media needed to preserve nucleic acid. If a lab requested PCR-quality testing, you should use the exact swab type and submission kit they provide (or ask the lab what to buy).
What should I do if I cannot get a bird’s mouth open to swab the oropharynx?
If you cannot access the oropharyngeal area safely, do not try to improvise with prolonged or rough handling at home. For flock or pen surveillance, some labs accept pooled oral fluid methods (for example, rope or sock exposure), but they still require correct material choice and the same transport and labeling standards. Ask the receiving lab whether they accept an alternative sampling method for your species and test.
Is it better to use BHI broth or viral transport medium (VTM/UTM)?
Either can be acceptable, but labs specify which one they want for a given test and sometimes the required volume. If you do not already know the target assay and acceptance criteria, contact the lab before collecting so you do not waste a sample in the wrong medium.
How long can the sample sit before shipping, and what if the cold chain breaks?
Quality depends on time and temperature. Short delays are best managed at 2 to 8°C, but if you expect longer than a few days, freezing at minus 70°C or below is usually needed. A broken cold chain can degrade PCR targets and lead to inconclusive or false-negative results, so when in doubt, tell the lab what happened and follow their re-collection guidance.
What happens if I pool more than the lab’s allowed number of swabs?
Pooling beyond the lab’s maximum (for example, more than five for some poultry surveillance workflows) can dilute the specimen enough to drop the viral load below detection thresholds. That increases false-negative risk, so pool only within the stated limit and with the same collection type and site.
Do I need to label whether the sample came from the beak/oropharynx versus cloaca?
Yes. Labs use the collection site to interpret results correctly and to choose the most appropriate testing approach for that specimen type. If you omit or mix sites without noting it, the lab may not be able to run the intended panel or may report the sample as not interpretable.
Are calcium alginate or foam swabs completely unusable?
Many diagnostic programs reject calcium alginate swabs because materials can interfere with downstream testing. Foam swabs can also be problematic depending on their composition. The most practical approach is to use synthetic, flocked or Dacron-style swabs on a plastic shaft, or follow the exact list of acceptable swabs from the lab you are submitting to.
Should I swab the trachea instead of the mouth if I want a stronger result?
Only in trained veterinary or properly supervised settings. Deeper airway sampling is more invasive and may require restraint and sometimes sedation, increasing risk to the bird and collector. For home collection, stick to the access approach you can do safely, and defer invasive sampling to a vet if a deeper sample is required.
If the bird is regurgitating or has visible discharge, can I collect that directly instead of swabbing?
You can capture visible secretions for basic observation, but for diagnostic testing you still need the correct specimen type and handling. Direct surface collection is more likely to be contaminated and may not preserve the pathogen or nucleic acid as well as a validated swab plus transport media. If you are aiming for lab PCR, confirm with the lab whether they accept that kind of specimen.
Do I need special packaging even if I’m just driving the sample to the vet?
Yes, at least for leak protection and contamination control. Use a sealed primary tube, place it in a secondary leak-proof container with absorbent material, and then keep it in a rigid outer container (triple-pack style). Even short-distance transport benefits from this because the outside of the tube is still considered potentially contaminated.
What personal protective equipment is most important if I’m collecting from birds at home?
Gloves and eye protection are key, because splashes can occur when birds flap or when you handle swabs near the beak. A mask is also appropriate, especially if birds appear respiratory ill or you suspect a higher-risk outbreak. Avoid relying on bare hands or open-tube handling to reduce exposure.
Can I test myself for exposure after a bird bite or contact with saliva?
Often the immediate health concern is assessed in the person, not by re-testing the bird’s saliva. Seek medical advice promptly if you have a bite or exposure, especially if the bird was wild or symptomatic. Bring details such as species, timing, and what contact occurred, and let clinicians decide whether any testing or prophylaxis is needed.




